Dr. John Schreiber Launches SCN8A Survey and Webinar for Medical Providers in Partnership with TCSF9/30/2019 In partnership with The Cute Syndrome Foundation, John M. Schreiber, MD from Children's National Hospital is collecting survey data from health care providers. The survey is intended to assess comfort in ordering and interpreting genetic testing and knowledge among medical professionals about SCN8A-related epilepsy, preferred treatments, and SUDEP risk. This research will help direct future education initiatives. Click the button below for access to the survey (Medical Professionals only, please). There is a also a free informational webinar given by Dr. Schreiber geared towards clinicians that will provide an overview of genetic evaluation in patients with refractory epilepsy with particular attention to SCN8A. This is being held by Global Genes from 1-2 pm EST on Wednesday, October 2. Please click on the following button to register. The password to access the webinar, when available is TCSF2019. This webinar will provide a brief overview of genetic evaluation in refractory epilepsy and specific examples of how a positive test may influence management. SCN8A-related epilepsy will be discussed in detail, with attention to epilepsy phenotype, diagnostic testing, associated co-morbidities including sudden unexpected death in epilepsy, and treatment.
0 Comments
Our SCN8A Annual Clinician, Researcher, and Family Gathering will be held in conjunction with the American Epilepsy Society Conference on December 6-7, 2019 at the Renaissance Harborplace Hotel in Baltimore Maryland. If you are a Medical Professional who would like to join the meeting or the caregiver of a child with SCN8A, please email Shelley Frappier for information about joining the event. The 2019 SCN8A Research Roundtable took place in Chapel Hill, North Carolina in July. This event brought together a select group of key SCN8A professionals for an intensive strategic planning meeting. Over a day and a half of meetings, participants focused on the state of SCN8A research, collaborate on grant efforts, and outlined next steps necessary to translate the scientific understanding of SCN8A into improved treatments and outcomes for individuals living with SCN8A. We believe the collaborative work between family advocacy groups, clinicians, researchers, and industry partners in driving research forward is key.
This year a very special team will be walking at the Walk to End Epilepsy hosted by Epilepsy Foundation of Metropolitan New York: Luke's Walk for Epilepsy.
Luke is one of the approximately 350 children in the world known to have SCN8A-related epilepsy--and his family is so eager to help find better treatment for Luke and the other children with SCN8A mutations. As a result, the Epilepsy Foundation of Metropolitan New York has generously agreed to donate half of the money Luke's team raises to The Cute Syndrome Foundation in support of SCN8A. Can you help them reach their goal of $50,000? Two weeks ago, Praxis Precision Medicines invited TCSF to their home base in Cambridge MA to talk to them about SCN8A community and to see how they can help—not just to help better treat SCN8A, but to do so as well as possible because they understand our experiences living with it.
Last week, Xenon announced that they had received positive feedback from the FDA regarding the development of XEN901 that supported advancing XEN901 directly into a pediatric clinical trial examining its efficacy in pediatric patients with SCN8A epileptic encephalopathy (SCN8A-EE). This is important as most Anti-Epileptic Drugs (AEDs) have to be studied in adults before they are studied in pediatric populations.
The Cute Syndrome Foundation is thrilled to share the awarding of a clinical research grant, originally announced at our 2018 SCN8A Clinician, Researcher, and Family Gathering, to Dr. John Schreiber and Children's National. The vision for this grant developed after the devastating loss of two infants in our SCN8A community last fall, and we are grateful to Dr. Schreiber for working with us to look for ways to improve access to expert advice in the treatment of individuals with SCN8A.
The Cute Syndrome Foundation is announcing the departure of Juliann Bradish, PharmD, as our Co-executive Director. Juliann will be remaining on as a volunteer at TCSF, turning her focus toward the administration of our support group. Juliann began volunteering for TCSF in 2015, serving as our SCN8A Advisor. She then joined Founder Hillary Savoie, PhD, as Co-executive Director in 2017. Juliann with her family at our 2016 SCN8A Clinician, Researcher, and Family Gathering
On November 30 and December 1st The Cute Syndrome Foundation held our fourth annual SCN8A Clinician, Researcher, and Family Gathering, in conjunction with the American Epilepsy Society meeting in New Orleans, LA. We were joined by more than 100 medical professionals, industry representatives, and family members as we presented two days of important activities—including discussions of SCN8A research, forthcoming potential therapeutics, and parent/patient perspective.
TCSF's 3rd Annual SCN8A Epilepsy Clinician, Researcher, and Family Gathering in Washington, D.C.12/22/2018 In December 2017 The Cute Syndrome Foundation hosted the third annual SCN8A Epilepsy Clinician, Researcher, and Family Gathering in Washington, DC.
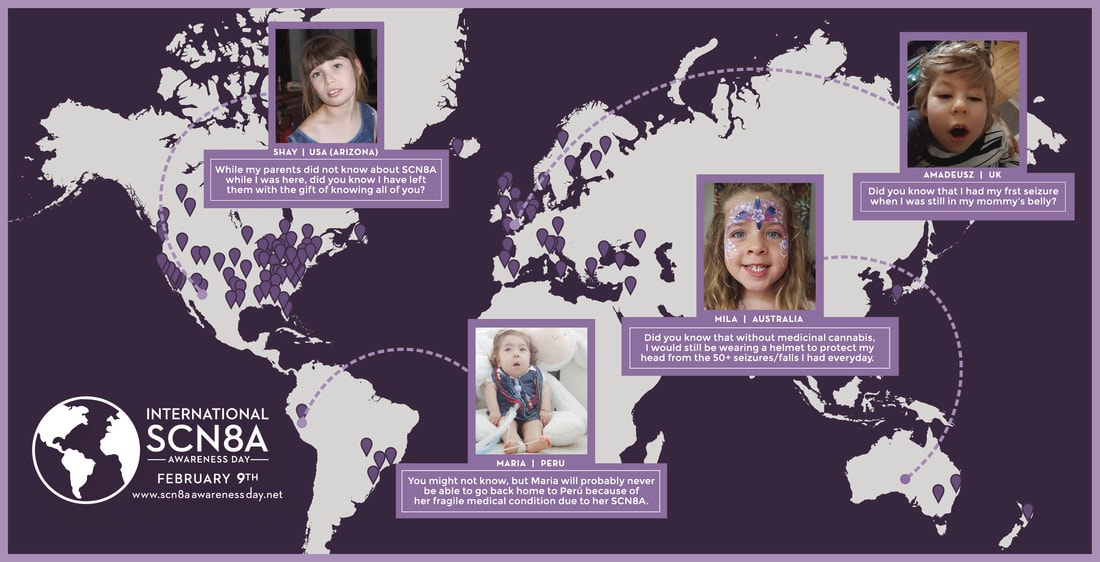
Over 100 guests met, including professionals working on SCN8A research and treatment and more than 75 members of our SCN8A family community, including 14 children with SCN8A. Families traveled internationally from Canada, Germany, Brazil, and New Zealand. The Cute Syndrome Foundation is celebrating the First International SCN8A Awareness Day on February 9th with the other family-based advocacy organizations including Ajude o Rafa and Wishes for Elliott, as well as individual families are reaching out to share the story of the grave impacts of this newly identified epilepsy. For more information about SCN8A Awareness Day visit: www.scn8aawarenessday.net
Play is a constant that still remains ever-changing, reinvented daily by kids and limited only by their boundless imaginations. But there is an artificial line between kids and adults when it comes to play. PLAE's goal is to erase that line and harness the power of play for everyone—starting with shredding the notion of age limits, of growing up, of losing that spirit of joy. On our journey, we start by asking ourselves, and the world, "Do you still PLAE?"
Every pair of PLAE shoes has been hand-crafted by a team of workers, with many pairs of hands adding their special touch to each pair that arrives at your door. Want to make it easier for TCSF to speed up the process of researching the SCN8A gene? Use checkout code GIVE494 every time you shop and we’ll donate 10% of your PLAE purchase price to The Cute Syndrome Foundation (TCSF). Shop now to earn a donation. The Cute Syndrome Foundation held our second annual SCN8A Epilepsy Research, Clinician, and Family Gathering on Friday December 2nd, in conjunction with the American Epilepsy Society Conference in Houston Texas. For the second year in a row we hosted over 100 SCN8A clinicians, researchers, and family members for an event built around sharing clinical information, research data, and family stories -- and the idea that these three groups can work together and help inform each other.
In January 2016, as a collaborative effort with our Brazilian partners, Ajude o Rafa, The Cute Syndrome Foundation awarded $25,000 the 2016 SCN8A Epilepsy Research Grant to Dr Miriam Meisler of The University of Michigan. On December 5th in Philadelphia about 85 guests--including seven children with SCN8A, 35 family members, and over 50 researchers and clinicians--met for the Cute Syndrome's first SCN8A Epilepsy Clinician, Researcher, and Family Gathering. The meeting allowed parents of children with SCN8A to tell leading researchers and clinicians more about their children and their lives. The meeting also served as a venue for clinical data about effective treatment of SCN8A to be shared among clinicians, and for researchers to share their research with the families--and each other.
The founder and director of the Cute Syndrome Foundation has published her second short book, titled Whoosh. Whoosh tells the story of a medical emergency that Esmé experienced at 3 months old. Read a short excerpt from the book here.
There is fantastic news for the treatment of PCDH19 Epilepsy. The University of Adelaide reports on the work of Prof. Josef Gecz, whose PCDH19 research has been supported with the help of our partner organization, Insieme per la Ricerca PCDH19 (Together for PCDH19 Research) in Italy. We are grateful that our partners recognized the potential for a promising drug treatment in Prof. Gecz's work and put their resources toward supporting him.
From the press release: An international team, led by a University of Adelaide genetics expert, has made a breakthrough discovery which is expected to help thousands of young girls worldwide who are suffering from a rare yet debilitating form of epilepsy. Professor Jozef Gecz, from the University of Adelaide’s Robinson Research Institute, was a key player in identifying the responsible gene and mutations in this female-only epileptic syndrome, in 2008. In breakthrough research published in Oxford Journals, Human Molecular Genetics, Professor Gecz has now found a treatment for this disorder. A United States pharmaceutical company Marinus Pharmaceuticals is now recruiting affected girls as part of the world’s first clinical trial to test the therapy. Insieme per la Ricerca PCDH19 and The Cute Syndrome Foundation are thrilled to announce that we have awarded a $20,000 grant to Dr. Jack Parent of the University of Michigan for his research using PCDH19 iPS cells. Dr Parent was a runner-up for our 2014 PCDH19 research grant (awarded to Dr. Maria Passafaro) and we are thrilled to support his research. It is our aim in 2015 to continue to support the most promising research proposals we received last year.
Hillary Savoie's writing was featured on Motherlode, the New York Time parenting blog. In her post on June 28th she reveals some wonderful news about her daughter Esmé: Esmé can read. Check out the post here: http://parenting.blogs.nytimes.com/2015/06/28/esme-can-read
Hillary's writing also has recently appeared on The Mighty and Complex Child Magazine. |
Archives
May 2024
Categories
All
|
|
Past Annual Reports
Past PCDH19 Efforts Privacy Policy The Cute Syndrome Foundation PO Box 842 Ozark, MO 65721 |
The Cute Syndrome is registered as a tax-exempt organization under IRS section 501(c)(3).
Our tax identification number is: 46-2699066. © The Cute Syndrome Foundation, All rights reserved |














 RSS Feed
RSS Feed


